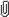source:
This is another area of interest to me as in how much memory do we become "prewired" either as a constant or an ability to reconstruct via our perception, right from day 1.
Developmental Profiles of Infant EEG: Overlap with Transient Cortical CircuitsExceprts:
Abstract and ObjectiveTo quantify spectral power in frequency specific bands and commonly observed types of bursting activities in the EEG during early human development.
Methods
An extensive archive of EEG data from human infants from 35 to 52 weeks postmenstrual age obtained in a prior multi-center study was analyzed using power spectrum analyses and a high frequency burst detection algorithm.
Results
Low frequency power increased with age; however, high frequency power decreased from 35 to 45 weeks. This unexpected decrease was largely attributable to a rapid decline in the number of high frequency bursts.
Conclusions
The decline in high frequency bursting activity overlaps with a developmental shift in GABA's actions on neurons from depolarizing to hyperpolarizing and the dissolution of the gap junction circuitry of the cortical subplate.
IntroductionFor decades pediatric neurologists have studied electrical signals recorded from the scalp of human infants. Initially, investigators sought to visually characterize patterns of electrical activity that were evident in these signals and to determine developmental changes in these patterns (Andre et al., 2010, Werner et al., 1977).
With the advent of improved computer technology, pioneers in the field used bioengineering methodologies to quantify the amount of electrical activity, i.e. power in a frequency-specific manner (Parmelee, 1969). These techniques revealed frequency dependent differences in power with sleep state (Curzi-Dascalova et al., 1988, Parmelee, 1969) and sleep position (Sahni et al., 2005), variance in power with clinical status (Scher, 1994), and differences between term and preterm infants (Scher et al., 1994a, Scher et al., 1994b).
Other investigators have characterized developmental changes in EEG during infancy, A notable study by Sterman and colleagues (Sterman et al., 1977) analyzed EEG data from birth to 24 weeks of age. They described a steady increase in low frequency power with PMA (Post Menstrual Age represents the age of the infant in weeks at discharge), while higher frequency power exhibited a “dip” from 40 to 44 weeks PMA.
Although not reported as a “dip”, data from Jenni et al. (Jenni et al., 2004) also showed a transient decrease in EEG power from 2 weeks after term birth to 2 months of age. Scher and colleagues found a transient decrease in high frequency power in both term and preterm infants and over a comparable period around 40 weeks PMA (Scher, 1997) and consistent with a report of a negative correlation between the 12-24 Hz beta of power and PMA from 36 to 43.5 weeks PMA in preterm infants (Scher et al., 1995). Although these data are suggestive of decreasing EEG power at higher frequencies through this early period of development, this pattern is not widely appreciated nor is there an understanding of what might account for this early decrease in high frequency EEG power.
To provide much-needed quantitative data of changes in the EEG during early development we analyzed an extant EEG data base collected during the
Collaborative Home Infant Monitoring Effort (CHIME). CHIME was an NICHD-funded multi-site study from the early to mid-90s designed to evaluate markers and risks for Sudden Infant Death Syndrome (SIDS) (Hunt, 1995). As part of this effort, overnight, ∼6 hour, polysomnographic studies were conducted with four channels of EEG in a large subset of infants. Although studied at a mean of 10 weeks after birth, the distribution of postnatal and postmenstrual ages was broad and encompassed the period of development from 35 to 52 weeks PMA.
For most of the polysomnograms, sleep states were coded in 30 sec blocks. Although many of the study subjects enrolled in CHIME were at increased risk for SIDS, we selected infants who were in the lower risk categories (i.e. healthy term and preterm infants without apnea and/or bradycardia near discharge) (Crowell et al., 2004, Crowell et al., 2002). Following this selection, there remained an adequate population of comparably studied infants for a robust cross-sectional study of changes in patterns of EEG power during early development.
For years, clinical electrophysiologists have recognized bursting activity in the EEG that is unique to early development. These bursts have been variously named slow or spontaneous activity transients (SATs), delta brush, and sharp transients (Andre, Lamblin, 2010, Biagioni et al., 1999, Biagioni et al., 2007, Hughes and Kohrman, 1989, Lamblin et al., 1999, Milh et al., 2007, Statz et al., 1982, Tolonen et al., 2007, Vanhatalo et al., 2005). These and other bursting patterns have been noted and quantified in EEG recordings in rodents (Seelke and Blumberg, 2008, Yang et al., 2009) and nonhuman primates (Myers et al., 1997, Myers et al., 1993, Stark et al., 1991).
Khazipov and Luhmann collectively termed these waveforms as “delta brushes”, apparent at ∼28weeks PMA and mostly gone by term age (Khazipov and Luhmann, 2006). Although generally thought to be of spontaneous origin, it has become evident that at least some bursts are linked to feedback from motor activity (Khazipov et al., 2004, Mohns and Blumberg, 2008). Although bursting activity in neuronal circuits is considered to be vital for normal cortical development (Ben-Ari et al., 2007) there is no consensus on exactly how bursts should be measured and categorized nor what their impact may be on traditional measures of EEG power.
The goal of this study was to quantify changes in power and bursting features of EEG activity unique to early development in order to develop novel methods for tracking specific neurophysiologic mechanisms critical for development of normal brain function. Our results substantiate previous inferences that EEG power above 10Hz does not increase from 34 to 44 weeks PMA as might be expected during this period of rapid brain growth.
Further, we find that the contributions to high frequency power attributable to short bursts of activity account for a significant amount of the total high frequency power and that these contributions decline steadily through this period. We discuss the potential linkage between our findings and the developmental shift in mechanisms underlying the ability of GABA to act as an inhibitory neurotransmitter, the dissolution of the circuits of the cortical subplate, and the progressive maturation of sleep states.
Infant PolysomnographyWiki (IPSG) and Sleep StateAn overnight polysomnographic recording was obtained for each subject at an average postnatal age (PNA) of 9.8 ± 5.8 (SD) weeks. The IPSG included EEG data from central (C3, C4) and occipital (O1, O2) electrode sites recorded with contralateral mastoid references (A1, A2). The CHIME acquisition hardware system (ALICE3) filtered the EEG data from 1 to 40 Hz with a notch filter at 60 Hz, and then digitized the signals with 8 bits per sample at the rate of 100 Hz. For a subset of subject population, CHIME investigators coded sleep state in 30-second epochs from autonomic data in the IPSG (Crowell et al., 1997, Hoppenbrouwers et al., 2005). For infants included in the current study, state data were coded for 174 infants (112 preterm, 62 term).
EEG Processing and AnalysisEEG data were processed in 30-second epochs to facilitate alignment with sleep state data. The root-mean-square (RMS) value of the EEG waveform for each 30-second epoch of data was computed and compared with a threshold to identify epochs containing artifact. Age-specific thresholds were set to define epochs of artifact that were apparent on visual review of the data (35-40wk, 35.8μV; 40-45wk, 39.8μV; and 45-55wk, 40.4μV). Most artifacts were associated with subject movements that were reflected in other IPSG data channels. The percentage of EEG data deemed as artifact varied with state and included 3% of data in quite sleep, 13% in active sleep.
To remove a potential for bias from the contralateral mastoid reference bipolar derivations were formed from the original 4 EEG channels. The bipolar channels were C3 to O1 on the left hemisphere and C4 to O2 on the right hemisphere. For each 30 second epoch of artifact-free data in these 2 channels, power spectra were computed. We used an FFT algorithm in custom MATLAB software that we had previously evaluated (Grieve et al., 2003). EEG power from the left and right hemispheres were computed and summarized as the mean of spectral values in 3Hz bands. For each 30 epoch, for each subject, EEG power was averaged within each of these 3Hz bands. Then, the average of these values was computed for epochs of quiet or active sleep. Outliers in power in each 3Hz band were defined as mean values exceeding 2 times the interquartile range above the 75th percentile or below the 25the percentile. These outliers were deleted prior to data analyses. In the worst case (19-21 Hz, left side), 4% of the data were excluded.
EEG Burst Detection and CharacterizationIn each 30 second epoch, high frequency bursts in EEG activity were detected and characterized with custom software following an algorithm depicted in Figure 1. For each 30 second epoch of EEG data (lower panel), burst detection was derived from results of spectral analysis of this waveform. Prior to the spectral analysis, the EEG was multiplied by a standard cosine on a pedestal window function that produced side lobes in the spectrum at least 40 dB below the main response (Harris, 1978). Then, the spectrum was calculated for each epoch (zero padded to 3032 points) using FFTs with a sliding 32 time-sample window which overlapped by one data sample and yielded a frequency resolution of ∼ 3Hz.
The resultant EEG spectral time series covered 0 to 40 Hz for each 30-sec epoch (middle panel). The highest frequency was limited to 40Hz by characteristics of the acquisition hardware. Next, for detection of high frequency bursts, the sum of power in the spectral time series between 10 and 40 Hz was smoothed with a 2 Hz low pass filter to eliminate EKG artifact (blue curve, upper panel).
A 10Hz high pass setting was adopted based on review of the literature describing high frequency oscillations in infant EEG (Milh, Kaminska, 2007, Tolonen, Palva, 2007). Next, for each epoch, a threshold for burst detection (red line, upper panel) was defined as two times the 10th percentile (green line, upper panel) of the smoothed 10-40Hz power time series. This threshold was arrived at by iteratively altering the threshold until the bursts captured by the automated method were in agreement with three experienced EEG analysts who concurred on visually identified high frequency bursts. Results of this algorithm for detecting high frequency bursts were consistent with those from visual inspection by multiple observers.
 Depiction of the burst detection algorithm to identify bursts of high frequency activity. The lower panel shows 30 sec of EEG and 4 high frequency bursts of varying duration (blue shaded areas). The middle panel shows the sliding window, 32 point FFT spectral time series from 0 to 40 Hz. Increasing amounts of power are depicted with a color scale from blue to red. The top panel shows the sum of power in the spectral time series between 10 and 40 Hz, smoothed with a 2 Hz low pass filter (black curve). The threshold for burst detection (red solid line) was defined as two times the 10th percentile (green dashed line) of the smoothed power. Each high frequency burst was characterized by peak amplitude, start and stop times corresponding to half peak power, and mean spectral frequency within the burst.DiscussionElectrical activity recorded on the scalp emanates from neuronal activity near the outer surface of the cortex and is due to synchronous postsynaptic activity
Depiction of the burst detection algorithm to identify bursts of high frequency activity. The lower panel shows 30 sec of EEG and 4 high frequency bursts of varying duration (blue shaded areas). The middle panel shows the sliding window, 32 point FFT spectral time series from 0 to 40 Hz. Increasing amounts of power are depicted with a color scale from blue to red. The top panel shows the sum of power in the spectral time series between 10 and 40 Hz, smoothed with a 2 Hz low pass filter (black curve). The threshold for burst detection (red solid line) was defined as two times the 10th percentile (green dashed line) of the smoothed power. Each high frequency burst was characterized by peak amplitude, start and stop times corresponding to half peak power, and mean spectral frequency within the burst.DiscussionElectrical activity recorded on the scalp emanates from neuronal activity near the outer surface of the cortex and is due to synchronous postsynaptic activity (Nunez and Srinivasan, 2006).
During the period over which the majority of the CHIME studies were performed (35-52 weeks PMA)
the human brain is growing rapidly in size and forming new circuits involving trillions of new synaptic connections (Khazipov and Luhmann, 2006).
Accordingly, increases in EEG activity (power) uniformly across frequencies through this period might be expected. Instead, we found frequency-specific changes in EEG activity that was dependent on sleep state.
In general, power below 7 Hz increased throughout the time period assessed while power from 7-9 Hz showed little change with age.
In contrast with these linear patterns, power in frequency bands above 9 Hz decreased from ∼36 to ∼45 weeks PMA and then remained relatively stable to 52 weeks (PMA).
An exception to this was power between 9 and 15 Hz which increased from ∼45 to 52 weeks (PMA) but only in QS.
The state dependent increase in 9 to 15 Hz power starting at 45 weeks, but only in QS, is likely due to the emergence of “sleep spindles” during this period (Jenni, Borbely, 2004). The combination of results reported in other studies is consistent with these diverse patterns of change in EEG power (Jenni, Borbely, 2004, Scher, 1997, Scher, Steppe, 1995, Sterman, Harper, 1977).
Our analyses of the CHIME data indicate that the observed decrease of high frequency power prior to term age is largely (74% in AS, 82% in QS) due to a several fold decrease in the frequency of occurrence of bursts of high frequency activity. While not widely recognized this developmental pattern may be understood in the context of the formation of transient microcircuits within the
cortical subplateWiki and their modulation by GABA
interneuronsWiki whose effects during this critical period of development transition from depolarizing to hyperpolarizing.
Following the early developmental concepts of Hebb (Hebb, 1949) many now accept the idea that
bursts of activity, whether spontaneous or evoked, are likely critical for circuit formation and subsequent brain development (Allene and Cossart, 2010, Ben-Ari, Gaiarsa, 2007, Katz and Shatz, 1996, Khazipov and Luhmann, 2006). Establishing the precise functions of these bursts and their neurophysiologic underpinning remains an important focus of current research. In the hippocampus of rodents, periodic increases in synchrony can lead to bursts known as giant depolarizing potentials (GDPs). These events can be of several hundred microvolts in amplitude and a few hundred milliseconds (Ben-Ari et al., 1989). Periodic enhancement of synchrony in neuronal firing also occurs in the neocortex and can be measured in the surface EEG (Seelke and Blumberg, 2008) and in cortical layers (Allene et al., 2008, Minlebaev et al., 2007, Rheims et al., 2008, Yang, Hanganu-Opatz, 2009).
GDPs in the hippocampus are seen at the same time during development that GABA has depolarizing effects. GDPs disappear with the transition in expression of chloride transporter-mediated inward (NKCC1) to outward (KCC2) flux and the transition in the effect of GABA to membrane hyperpolarization (Ben-Ari, Gaiarsa, 2007, Rheims, Minlebaev, 2008). Consistent with these findings, bumetanide, a selective inhibitor of NKCC1, blocks GDP in slices during the early postnatal period (Dzhala et al., 2005). Bumetanide also blocks sharp waves in the rat hippocampus at postnatal days 7-9 which are thought to be the in vivo version GDPs (Sipila et al., 2006).
The idea that the transient developmental expression of some types of cortical bursting patterns in EEG might similarly be related to the early depolarizing effects of GABA was first suggested by Vanhatalo and colleagues (Vanhatalo, Palva, 2005). This group described and quantified bursts of activity that they called
Spontaneous Activity Transients (SATs) identified primarily by their high amplitude (∼100uV) and low frequency content. It is important to note that these transients also contain power across a broad frequency spectrum including high frequencies (Tolonen, Palva, 2007). The incidence of SATs declines dramatically during the perinatal period, disappearing by a few weeks after birth (Tolonen, Palva, 2007, Vanhatalo and Kaila, 2006, Vanhatalo, Palva, 2005).
A similar decline in SAT-like features was reported in the EEG of rats during the early postnatal period (Seelke and Blumberg, 2008). Analyses of postmortem human infant cortical tissue obtained at several ages through this period demonstrated a transition in expression of chloride transporters from the inwardly transporting NKCC1 to the outwardly transporting KCC2 (Dzhala, Talos, 2005). Vanhatalo and colleagues noted that the decline in SATs parallels the transition in the effects of GABA from depolarizing to hyperpolarizing (Vanhatalo, Palva, 2005), thus the conclusion that cortical SATs may be dependent on the depolarizing effects of GABA.
The burst detection method we used quantifies all high frequency bursts, not only those coincident with low frequency bursts of activity (e.g. SATs and delta brush). We identified bursts of many types. Vanhatalo and colleagues have shown that even though SATs contain high frequency power (Tolonen, Palva, 2007) precise detection of SATs requires that the EEG be recorded with DC amplifiers that allow low frequency transients to be seen (Vanhatalo and Kaila, 2008). However, CHIME data were high pass filtered at 1 Hz which would impede the accurate detection of onset and offset of SATs.
Thus, we cannot determine the proportion of high frequency bursts measured by our algorithm that are associated with SATs. Moreover, the minimal spatial resolution (2 bipolar leads) and low sampling rate (100Hz) used in CHIME preclude determination of the distribution of bursts over the cortical surface and whether there are bursts at higher frequencies. In addition, we are unable to quantify bursting activity prior to 35 weeks PMA.
Future studies should acquire data at earlier ages, address topographic distribution of these developmental patterns, and assess frequencies above 24 Hz to enrich these analyses. Despite the limitations of the CHIME data set, we show that high frequency bursts decline with age (see Figure 6) over the same time course as others found SATs decline (Tolonen, Palva, 2007, Vanhatalo and Kaila, 2006, Vanhatalo, Palva, 2005). Albeit speculative, this suggests that high frequency bursts might be used as a marker for the transitions in the effects of GABA on membrane polarization.
The speculation that bursts of EEG activity of various forms may be linked to the depolarizing actions of GABA during early stages of brain development is paralleled by
an hypothesis that relates the bursting activity to a cortical structure that, like neuronal responses to GABA, undergoes rapid changes during the late prenatal and early postnatal period.
This structure is the layer of neurons and interneurons referred to as the subplate. The subplate lies beneath the primitive cortical plate and, at its maximum size, which is achieved in humans between 22 and 34 weeks PMA, is several fold thicker than the cortical plate (Kanold and Luhmann, 2010, Kostovic et al., 1989, Kostovic and Rakic, 1990). The subplate circuitry is formed by interconnections between neurons in the subplate and cortical plate, from ascending thalamic fibers, from GABAergic interneurons, and from ascending monoaminergic fibers (Kanold and Luhmann, 2010, Kostovic and Rakic, 1980, 1990, Luskin and Shatz, 1985, Weber and Andrade, 2010).
Numerous studies have shown that the subplate neural circuitry is required for normal cortical development (Allendoerfer and Shatz, 1994, Clowry et al., 2010, Ghosh et al., 1990, Ghosh and Shatz, 1992, Kanold et al., 2003, Soriano et al., 1994, Wood et al., 1992, Xie et al., 2002). Of particular interest to the current findings, many studies have shown that subplate circuits are transient and in humans regress during the last weeks of gestation and early postnatal period although in some regions these circuits may be sustained for up to 6 months of age or longer (Delalle et al., 1997, Kanold and Luhmann, 2010, Kostovic and Judas, 2007).
Although there are no in vivo studies in the subplate, the role of this transient layer in generation of high frequency bursts is supported by electrophysiological studies of early postnatal mouse cortical tissue slices. In one study, brief (2 msec) electrical stimulation of the subplate resulted in high frequency (∼18Hz) oscillations in the cortical plate lasting several hundred msec (Sun and Luhmann, 2007). In another study, also conducted in mice, application of cholinergic agonists to cortical slices produced bursts of activity at a comparable frequency range (∼17Hz) (Dupont et al., 2006).
Although these cholinergic-induced bursts were not blocked by a GABA-A antagonist, this study did show that these high frequency bursts were dependent on gap junction based circuitry of the subplate but only up to about 3 days of age. In subsequent studies, it was found that activation by non-synaptic release of GABA facilitates the generation of “up states” during which bursting activity is produced and prolongs cholinergic-induced depolarization of subplate neurons (Hanganu et al., 2009). These findings in mice are of interest in relation to the current study because the frequencies within these cholinergic-induced bursts are essentially the same as the mean spectral frequency of the spontaneous bursts we identified in sleeping infants (17.3Hz in QS, 18.5Hz in AS).
The decrease in bursting activity we quantified in this current study occur over the time interval expected for the transition in GABA effects on membrane polarization and with the dissolution of the cortical subplate. However, these are only temporal associations and do not demonstrate causal, mechanistic links. Other researchers have reported that in both preterm human infants and early postnatal mice high frequency bursting activity in response to visual stimulation.
This response disappears with age and is replaced by the typical evoked response seen later in development. These light-induced bursts are not blocked by a GABA-A antagonist (Colonnese et al., 2010) and therefore not dependent on the depolarizing effects of GABA expected during this period of development. These authors also proposed that the disappearance of these bursts was not related to the disappearance of the subplate but no direct evidence of this was shown. It is important to note that the transition in the response to light flashes in human infants occurred from 34 to 36 weeks, prior to the disappearance of bursting activity we quantified. Thus, light-induced bursts and the spontaneous bursting activity we recorded differ in development course and are not likely to be related.
In other studies Marcano-Reik and colleagues showed that in rat pups before 6 days of age high frequency bursting activity in the somatosensory cortex associated with myoclonic twitches is increased following administration of bicuculline, a GABA-A antagonist (Marcano-Reik et al., 2010). Transection of the corpus callosum increased the number of bursts associated with twitches without altering the number of twitches. This increase in bursting activity was also potentiated by
bicucullineWiki.
These findings are discordant with the hypothesis that depolarizing effects of GABA are permissive if not casually related to bursts since antagonism of GABA-A receptors did not inhibit bursts. These authors proposed that although the effects of GABA on membrane potential at this early age might be depolarizing, the major effect of GABA is nonetheless inhibitory due to shunting. However, it is not clear whether the inhibitory effects of GABA on twitch-related bursts in rats applies to the multiple types of bursting activity we characterized in human infants.
The results provided in Table 2 show an addition finding from this study. Power below 13 Hz is greater in the right hemisphere, but only at the earlier PMA age range (35 to 45 wks). This is interesting in light of findings showing auditory evoked responses have shorter latencies on the right side in fetuses 29 to 38 wks of age (Schleussner et al., 2004), and greater amplitude on the right side in infants born between 30 and 34 wks GA and studied at about 35 PMA (Mento et al., 2010). These data suggest that the right hemisphere may develop more rapidly than the left, and our results provide evidence that resting EEG power may also be a marker of this asymmetry in maturation. We did not find a left/right difference in the rate of bursting through this age range. However, bursts as we measured them are higher frequency phenomena and thus may reflect a different aspect of cortical maturation than low frequency power.
Although developmental timing is not precise, the process of subplate dissolution overlaps with the transition in chloride transporter gene expression and the switch in GABA effects on membrane polarization. It is not clear if these two development phenomena are mechanistically linked. We propose that bursting activity measured in the EEG of premature infants requires intact transient subplate circuits and their connections to the cortical plate. In addition, some types of bursting activity may be related to membrane depolarizing effects of GABA; although, this may not be true for all types of bursts.
Disruptions in GABA's actions and subplate function during early development cause structural and functional changes that could underlie dysfunction in sensory processing, emotion regulation, and cognition which are apparent in a wide range of mental disorders including autism and schizophrenia. (Avino and Hutsler, 2010, Ferriero and Miller, 2010, Kostovic et al., 2011, Wang and Kriegstein, 2011). Currently, it is not possible to determine in human infants the progression of these fundamental neurophysiologic processes. Our analyses of the CHIME data set are informative with regard to helping establish norms for EEG development and suggest that systematic quantitative analyses of resting EEG activity can provide a means of tracking specific neurophysiologic mechanisms that are key to normal brain development.