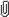2.3.
Phenibut Referred scheduling proposalA delegate from the Therapeutic Goods Administration has referred a scheduling proposal to create a new Schedule 9 or Schedule 4 with an Appendix D, Part 5 entry for phenibut in the Poisons Standard. Scheduling application This was a delegate initiated application.
The delegate’s proposed amendments to the Poisons Standard are: Schedule 9 – New Entry PHENIBUT. OR Schedule 4 –
New Entry PHENIBUT. Appendix D, Part 5 – New Entry PHENIBUT. AND Index –
New Entry PHENIBUT cross reference:
BETA-PHENYL-GAMMA-AMINOBUTYRIC ACID Schedule 4/9 Appendix D, Part 1
The reasons for the request are:
• Case reports of significant toxicity have emerged, as well as evidence of dependence; and
• One state has raised concerns regarding the potential for tolerance and withdrawal symptoms from the use of Phenibut.
Specific toxicity is related to these symptoms. Current scheduling status and relevant scheduling history Phenibut is currently not captured by any schedule entry in the current Poisons Standard.
The pharmacologically similar substance, Baclofen, is in Schedule 4 and Appendix K of the current Poisons Standard. Phenibut has not been previously considered for scheduling. Therefore, a scheduling history is not available. Australian regulatory information Phenibut is not listed as an ingredient in products on the ARTG and cannot be legally sold in Australia as a Therapeutic Good.
However, information received from one Australian state health department indicates that phenibut is marketed to relieve anxiety and depression, improve sleep and enhance cognition. Despite phenibut not being in products on the ARTG, the Database of Adverse Events Notification (DAEN) - Medicines has returned 1 report of an adverse event suspected to be related to phenibut in an unregistered product. Phenibut is not listed in the Therapeutic Goods (Permissible Ingredients) Determination No. 3 of 2017.
International regulations No evidence of scheduling in New Zealand, EMA, US FDA or Canada. Substance summary Phenibut is a neuropsychotropic drug with anxiolytic and nootropic (cognition enhancing) effects.
It acts as a GABA mimetic, primarily at GABAB and to some extent at GABAA receptors.
Table 2.3.1: Chemical information for phenibut Property Phenibut Chemical structure Molecular formula C 10 H 13 NO 2 Molecular weight 179.2 CAS number 1078g/mol 213 Delegates’ interim decisions and reasons for decisions September 2017.
Eleven (11) public submissions were received, two (2) in support, eight (

opposed (four (4) to the Schedule 9 proposal and four (4) showing some agreement with a Schedule 4 entry to allow access via a prescription) and one (1) did not state their position.
Main points in support:
• There are no established therapeutic uses for phenibut.
• In Australia there are confirmed cases of phenibut poisoning and an increase in the number of suspected cases of phenibut use, misuse and harm.
• Phenibut is marketed on the internet as a dietary supplement to treat anxiety and sleep disorders. However, there is strong evidence to indicate that it is predominantly used as a recreational drug. • The medical conditions phenibut is reportedly being used to treat (including anxiety and sleep disorders) are better managed by a medical practitioner.
• Phenibut represents a significant risk of harm, including overdose (intentional and accidental). Complications of overdose include coma requiring admission to an Intensive Care Unit (ICU) for advanced life support. • Withdrawal symptoms result when phenibut is stopped.
• No preference between Schedule 4/Appendix D or Schedule 9 was indicated in the submissions. Main points opposed:
• Personal stories indicate that where other medicines have failed phenibut has: – provided improvement to sleep and symptoms of anxiety, depression, idiopathic hypersomnia and Post-Traumatic Stress Disorder (PTSD); and – been used to treat addiction to alcohol and benzodiazepine use. • Claims that suppliers of phenibut provide adequate information on its safe use.
• Consumers have had no problems with toxicity or dependence and believe phenibut is very safe. • Scheduling phenibut may cause an increase in alcohol consumption, use of stronger anti-anxiety medicines or black-market drug purchasing.
• Internationally phenibut is only regulated in Russia. No evidence for scheduling in other countries.
• Submissions indicate some preference for: – Schedule 4 without an appendix entry to allow access via a prescription; and – Schedule 3 to allow access by a pharmacist. The public submissions will be made available on the TGA website.
Summary of ACCS-ACMS advice to the delegate The committee recommended that new Schedule 9 and index entries be created in the Poisons Standard for phenibut as follows: Schedule 9 – New Entry PHENIBUT. Index – New Entry PHENIBUT cross reference: BETA-PHENYL-GAMMA-AMINOBUTYRIC ACID Schedule 9 The committee also recommended an implementation date of 1 February 2018. Members agreed that the relevant matters under Section 52E(1) of the Therapeutic Goods Act 1989 included:
(a) risks and benefits of the use of a substance;
(b) the purpose for which a substance is to be used and the and extent of use;
(c) the toxicity of a substance;
(d) the dosage, formulation, labelling, packaging and presentation of a substance;
(e) the potential for abuse of a substance; and
(f) any other matters that the Secretary considers necessary to protect public health.
The reasons for the advice were:
• Risks of phenibut include tolerance, dependence, abuse, accidental and intentional overdose resulting in significant toxicity requiring hospitalisation, with severity potentially requiring ICU admission.
• There are anecdotal reports of the therapeutic benefit of phenibut based on public submissions. • Although phenibut is used therapeutically in Russia, the associated clinical trial literature is unable to be evaluated critically at this time due to translation issues. Furthermore, there has been no established therapeutic benefit of phenibut in regulatory comparable countries.
• Taking into consideration the danger to the health of individuals and of the community (both immediate and imminent) associated with the use of phenibut and the high risk of dependency, abuse, misuse and illicit, the perceived benefits (as indicated in public submissions) are substantially outweighed by the risks.
• The substance is not currently permitted in Australia to be marketed for therapeutic reasons but is widely available on the internet for purchase. International websites make significant therapeutic claims for cognition enhancement, anxiety and depressive disorders. Prevalence of use is not established but is clearly across the country and increasing.
• P
henibut is a neuropsychotropic drug with anxiolytic and cognition enhancing effects. It acts as a GABA mimetic, primarily at GABAB and to some extent at GABAA receptors. Published reports of ED presentations, acute intoxication with delirium, and dependence treated with baclofen. These reports include reports from Australia. Descriptions of withdrawal include tremors, anxiety, insomnia, hypertension, hyperhidrosis, psychosis, tachycardia, widening of QRS complex and convulsions. There is significant risk of harm. Effects include CNS depression, delirium, seizures – potentially requiring intubation and ventilation.
• Rapid development of tolerance and dependence with a withdrawal syndrome consisting of hallucinations, agitation, tremor, insomnia, abdominal pain, vomiting.
• The availability as a powder increases risks of toxicity.
Delegate’s considerations The delegate considered the following regarding this proposal:
• Scheduling proposal
• ACCS-ACMS advice • Public Submissions received
• Section 52E of the Therapeutic Goods Act 1989
• Scheduling Policy Framework (SPF 2015)
• Other relevant information Delegate’s interim decision The delegate’s interim decision is to create new Schedule 9 and index entry in the Poisons Standard for phenibut.
The proposed Schedule entry is as follows: Schedule 9 – New Entry PHENIBUT. Index –
New Entry PHENIBUT cross reference: BETA-PHENYL-GAMMA-AMINOBUTYRIC ACID Schedule 9.The proposed
implementation date is 1 February 2018 as this is the earliest possible implementation date.[/color]
The matters under subsection 52E(1) of the Therapeutic Goods Act 1989 considered relevant by the delegate included:
(a) the risks and benefits of the use of a substance;
(b) the purposes for which a substance is to be used and the extent of use of a substance;
(c) the toxicity of a substance;
(d) the dosage, formulation, labelling, packaging and presentation of a substance;
(e) the potential for abuse of a substance; and
(f) any other matters that the Secretary considers necessary to protect public health.
The reasons for the interim decision are:
(a) the risks and benefits of the use of a substance:
• There are anecdotal reports of the therapeutic benefit of phenibut based on public submissions.
• Although phenibut is used therapeutically in Russia, the associated clinical trial literature is unable to be evaluated critically at this time due to translation issues. Furthermore, there has been no established therapeutic benefit of phenibut in regulatory comparable countries.
• International websites make significant therapeutic claims for cognition enhancement, anxiety and depressive disorders.
• Risks of phenibut include tolerance, dependence, abuse, accidental and intentional overdose resulting in significant toxicity requiring hospitalisation, with severity potentially requiring ICU admission.
• The perceived benefits (as indicated in public submissions) are substantially outweighed by the risks.
(b) the purposes for which a substance is to be used and the extent of use of a subalance.
The substance is not currently permitted in Australia to be marketed for therapeutic reasons but is widely available on the internet for purchase. International websites make significant therapeutic claims for cognition enhancement, anxiety and depressive disorders. Prevalence of use is not established but is clearly across the country and increasing.
• Prevalence of use is not established but is clearly across the country and increasing.
(c) the toxicity of a substance:
• Phenibut is a neuropsychotropic drug with anxiolytic and cognition enhancing effects. It acts as a GABA mimetic, primarily at GABAB and to some extent at GABAA receptors.
• Published reports of ED presentations, acute intoxication with delirium, and dependence treated with baclofen. These reports include reports from Australia.
• Descriptions of withdrawal include tremors, anxiety, insomnia, hypertension, hyperhidrosis, psychosis, tachycardia, widening of QRS complex and convulsions. There is significant risk of harm.
• Effects include CNS depression, delirium, seizures – potentially requiring intubation and ventilation.
• The availability as a powder increases risks of toxicity.
(d) the dosage, formulation, labelling, packaging and presentation of a substance:
• Packaged as a supplement in 250 and 500mg capsules.
• No restrictions at present, currently available in powder and capsules with variable labelling.
• The availability as a powder increases risks of toxicity. Loose powders pose particular risk of accidental overdose.
(e) the potential for abuse of a substance:
• Rapid development of tolerance and dependence with a withdrawal syndrome consisting of hallucinations, agitation, tremor, insomnia, abdominal pain, vomiting.
• Cases of recreational abuse have been described.
• Rapid development of tolerance is established.
(f) any other matters that the Secretary considers necessary to protect public health:
• Taking into consideration the danger to the health of individuals and of the community (both immediate and imminent) associated with the use of phenibut and the high risk of dependency, abuse, misuse and illicit, the perceived benefits (as indicated in public submissions) are substantially outweighed by the risks.